BACKGROUND:
Chimeric antigen receptor (CAR) T-cell therapy has changed the treatment landscape for B-cell non-Hodgkin lymphoma (B-NHL). Results from recent studies have expanded patient access to this treatment. However, little data is available evaluating the impact of time from referral to treatment on outcomes or cost for patients receiving CAR-T.
METHODS:
Patients (pts) aged ≥18 years with B-NHL who were referred either internally or externally between November 2017 and March 2023 for commercial CAR-T at Vanderbilt University Medical Center were included. Pts who died prior to leukapheresis or infusion were not excluded. Pts on a clinical trial were excluded. Date of referral was defined by presentation at an internal consensus meeting shortly after the initial provider visit. Other key time points were date of leukapheresis and date of CAR-T infusion.
Cox regression analysis and the Kaplan Meier method were used for survival outcomes. Results were stratified by time to leukapheresis (within vs. greater than 45 days) as a proxy for a reasonable timeline, including evaluation, insurance approval, line placement, and scheduling. Pts who died prior to leukapheresis were included in the greater than 45 day group. Post referral therapy included treatment given between referral and leukapheresis, and bridging therapy included treatment given after leukapheresis.
A crude cost estimate of therapies prior to CAR-T infusion was performed accounting for drug costs by type and number of cycles, administration (inpatient vs. outpatient), and estimated related costs of complications. Dollar amounts were obtained by review of CMS and previously published studies. Mann-Whitney U test was used to compare mean cost differences.
RESULTS:
Baseline characteristics are shown in Table 1. A total of 51 pts received axicabtagene ciloleucel (axi-cel, 78%), tisagenlecleucel (tisa-cel, 7%), or brexucabtagene autoleucel (brexu-cel, 15%) with a median follow up of 280 days. There were 9 pts (15%) who died while awaiting leukapheresis. The median age at referral was 62 (range 22-84). More pts had relapsed (43%) disease than relapsed/refractory (22%) or primary refractory disease (35%). Most patients had stage 3 or 4 disease (72%) and elevated LDH (75%). Private insurance (63%) was more common than Medicare (37%).
The median time from referral to leukapheresis was 67 days (range 21-182) and median time from referral to CAR-T infusion was 99 days (range 49-212). There were 43 pts (73%) who underwent leukapheresis after 45 days. Most pts (85%) required post-referral therapy and many pts (21%) required two or more lines. Post referral therapy regimens were platinum-based (39%), polatuzumab-based (29%), other systemic chemotherapy (16%), and oral targeted agents (16%). Few pts (35%) required bridging therapy after leukapheresis with only two pts getting steroid monotherapy as bridging.
The 12-month overall (OS) and progression free (PFS) survival for the cohort were 59.3% and 54.5% respectively. There was no difference in survival outcomes when stratified by early vs. late leukapheresis. Time from referral to leukapheresis or CAR-T infusion were not associated with survival outcomes in multivariate analysis.
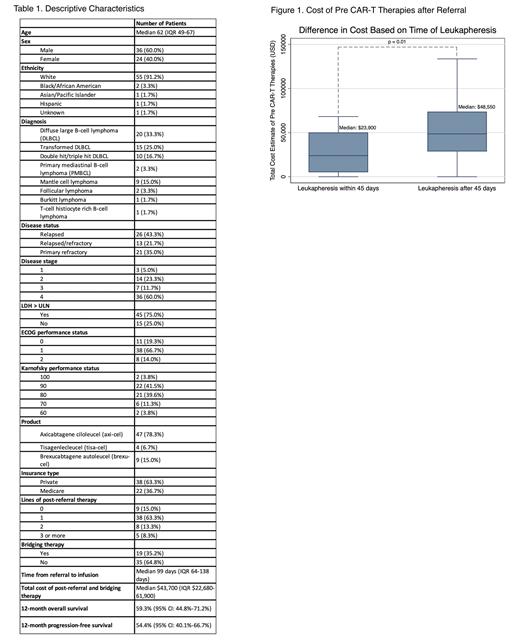
Excluding costs for leukapheresis and CAR-T infusion, the median cost of post-referral and bridging therapies was $43,700 USD (range $22,680 - $133,570). The median cost of therapies (Figure 1) for pts who underwent early leukapheresis was lower than for pts who underwent late leukapheresis (median $23,900 vs. $48,550, p=0.01). In a multivariate regression analysis, time from referral to leukapheresis (p=0.008) and the receipt of bridging therapy (p=0.010) were associated with increased costs. A sensitivity analysis performed based on year of referral did not change survival or cost outcomes.
CONCLUSION:
In this intention to treat analysis, we showed that time from referral to CAR-T infusion is longer than expected but did not impact survival in multivariate analysis. Prior to leukapheresis, most pts required one or more lines of systemic therapy. Given increased healthcare utilization, the cost of therapies prior to CAR-T was high, reflecting a pressing need to minimize logistic barriers prior to CAR-T to reduce costs and improve patient quality of life. Future multicenter studies should be conducted to better understand the impact of time from referral to treatment on costs, outcomes, and quality of life.
Disclosures
Dholaria:Gilead: Research Funding; BEAM therapeutics: Consultancy; Adicet: Research Funding; ADC therapeutics: Consultancy, Honoraria; Angiocrine: Research Funding; Takeda: Research Funding; Wugen: Research Funding; Arivan: Consultancy; Pfizer: Research Funding; MEI: Research Funding; Orca Bio: Research Funding; Poseida: Research Funding; Allovir: Research Funding; NCI: Research Funding; Atara: Research Funding; Molecular Templates: Research Funding; AstraZeneca: Research Funding; Pluri Biotech: Consultancy; Boxer Capital: Consultancy; Ellipsis pharma: Consultancy; Lumanity: Consultancy; gamida cel: Consultancy; Janssen: Consultancy, Honoraria, Research Funding; BMS: Research Funding; Poseida: Research Funding. Oluwole:Novartis: Consultancy; Nektar: Consultancy; Kite, a Gilead Company/ Gilead: Consultancy, Research Funding; Epizyme: Consultancy; Caribou: Consultancy; Cargo: Consultancy; ADC: Consultancy, Speakers Bureau; AbbVie: Consultancy; Pfizer: Consultancy, Honoraria, Research Funding; Gilead: Consultancy, Honoraria; TGR: Consultancy; Allogene: Research Funding; Daiichi Sankyo: Research Funding. Savani:Takeda Development Center Americas, Inc. (TDCA): Current Employment. Sengsayadeth:Amgen: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal